Hip implants Knee implants Spinal implants Shoulder implants Finger implants Osteosynthesis Instruments Ankle implants Vascular implants Material testing Dental implants Particle analysis Elbow implants Wrist implants Exoprosthesis Consumables
Implant Testing
Our laboratories are performing mechanical tests for almost every type of implant.
Pre-clinical implant testing has become one of the most important tools for safety analysis of medical devices. A vast number of publications on clinical performance and biomechanical testing as well as national and international test standards are available to develop the optimal test protocol for each individual product.
EndoLab® - Implant Testing Services Brochure [PDF]
EndoLab® is closely cooperating with standardization organizations such as
ASTM and ISO to improve current testing standards and develop future implant tests.
Our clients benefit from modern test equipment and highly innovative test methods.
EndoLab® does develop new and highly innovative test equipment for implant testing such as
multi station hip and knee simulator wear test frames. We also provide testing services for
vascular implants and instruments such as pen injectors.
Our test labs do offer a variety of material tests such as physical testing (compression, tension and shear) and characterization of coatings. EndoLab® does perform
particle analysis and ion release tests for any type of modular implant. We do run one of the leading labs for
corrosion testing and fretting corrosion analysis. As there are specialists for all aspects of implant testing available, your testing needs will be covered most efficiently.
Of course, all tests are performed under our stringent quality system according to ISO 17025. Our clients benefit from our
accredited test procedures and up to date implant testing process. The EndoLab® database will support your certification process and guarantee an optimized testing period.
Being in the business for over 20 years, we are here to help you define your specific implant testing needs. Our experience in the development and validation of new test methods will support your R&D team in the design process.
Our
large scope of certification will cover almost any implant testing requirement.
Contact EndoLab® today to learn more about our implant testing service.
 Implant testing at EndoLab GmbH (©2024)
Implant testing at EndoLab GmbH (©2024)
 EndoLab® is one of the leading companies in international implant testing
EndoLab® is one of the leading companies in international implant testing
 More than 20 years experience in development, certification (CE and FDA) and marketing of medical products
More than 20 years experience in development, certification (CE and FDA) and marketing of medical products
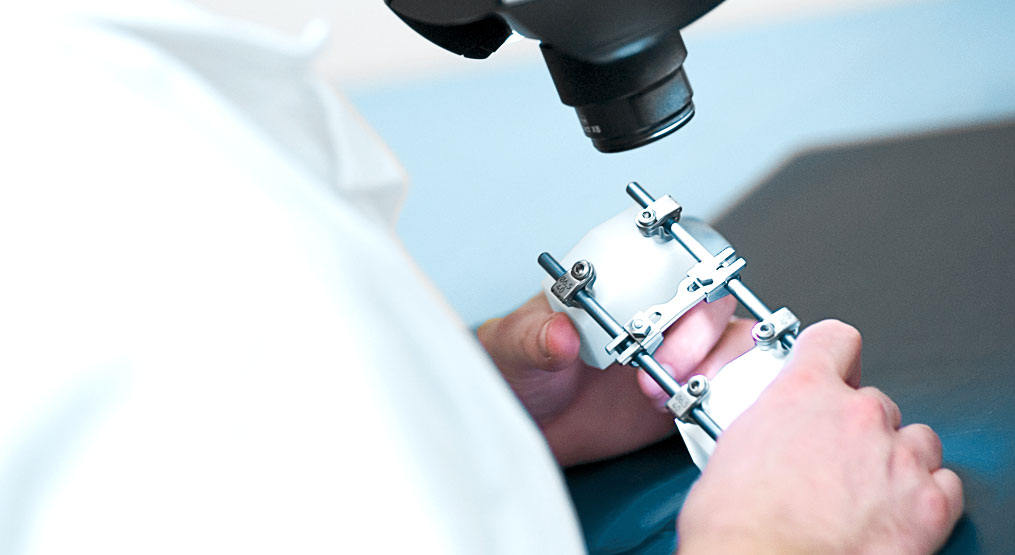 ISO 17025 certified test laboratory for medical implant testing and material tests
ISO 17025 certified test laboratory for medical implant testing and material tests

